Insulin is one of the most widely produced biopharmaceuticals in the world. With hundreds of millions of people dependent on it to manage diabetes, the reliability and precision of every step in its manufacturing chain is critical. Among those steps, mixing is often overlooked, yet it is where product quality, protein integrity, and process efficiency are determined.
Why Is Mixing So Critical in Insulin Production?
The challenge lies in the final preparation stages. Insulin must be injected directly into the bloodstream, so each vial must contain a highly precise concentration. Preparing dosage solutions requires thorough mixing across large volumes, while maintaining the integrity of a protein that is extremely sensitive to mechanical stress. Exposure to intense turbulence triggers aggregation, which progresses from early-stage clustering to irreversible fibrillation. Once denatured, the protein cannot be recovered. The result is failed quality controls and lost batches.
The FUNDAMIX® Vibro-Mixer from DrM Group addresses this challenge directly. Its electromagnetic vibration technology delivers thorough, homogeneous mixing without the mechanical stress, sterility risks, or cleaning complexity associated with conventional stirred tank agitators. More than 580 FUNDAMIX® units have been installed across pharmaceutical and biotechnology facilities in 41 countries, with insulin production among its most established applications.
How Does FUNDAMIX® Solve the Problem?
Conventional stirred tank agitators create intense turbulence zones near the rotating impeller tip. Even at moderate speeds, these zones are sufficient to initiate aggregation in sensitive proteins. The faster the impeller must rotate to achieve homogenization, the greater the risk to the product. The FUNDAMIX® Vibro-Mixer eliminates this trade-off entirely.
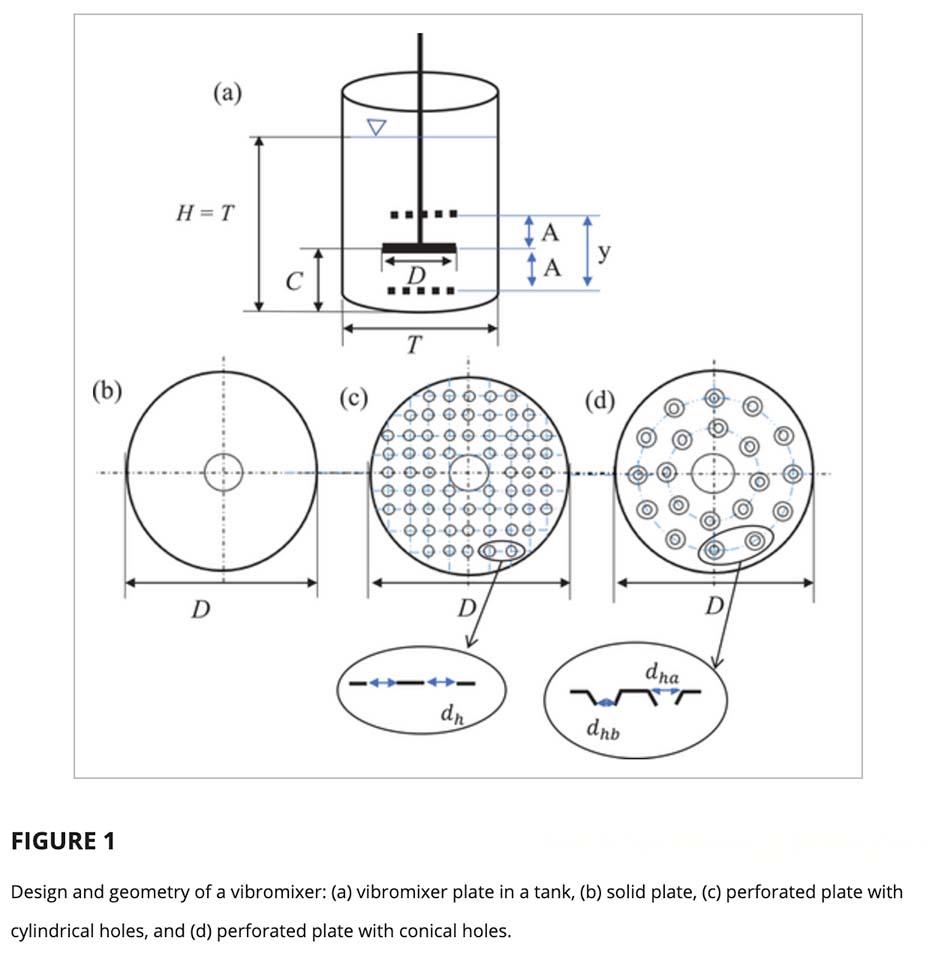
The FUNDAMIX® Vibro-Mixer operates on a fundamentally different principle. An electromagnetic drive unit oscillates a perforated mixing plate vertically at controlled frequency and amplitude. This generates axial flow through the vessel without any rotating components entering the product. The mixing intensity can be adjusted in real time by varying frequency and amplitude independently, giving operators precise control over the process without stopping or disassembling the system.
The result is thorough, homogeneous mixing at a fraction of the specific power input of a rotating impeller, and with a turbulent Reynolds number well below the threshold at which protein damage occurs:
| Parameter | FUNDAMIX® Vibro-Mixer | Rotating Impeller |
|---|---|---|
| Specific power (W/m³) | 510 | 8,600 |
| Dimensionless mixing time | 1.5 to 3 | 10 to 100 |
| Turbulent Reynolds number | 20 to 300 | > 10,000 |
Gimba et al., Chem. Eng. & Tech., 2026. https://onlinelibrary.wiley.com/doi/10.1002/ceat.70149
What Does This Mean for GMP Pharmaceutical Production?
Beyond protein protection, insulin manufacturers need mixing equipment that fits cleanly into a validated, cGMP production environment. FUNDAMIX® is built for exactly that.
All product-contact surfaces are CIP (Clean-In-Place) and SIP (Sterilize-In-Place) compatible. The membrane sealing unit carries FDA, USP Class VI, and EN 10204 3.1 certification. The drive unit comes with standard IP65 protection, with an ATEX option available. Due to the absence of rotating parts, there is no need for complex mechanical seals or vortex breakers and baffles that complicate cleaning validation, resulting in a simpler validation package and faster time to qualification.
The system scales from laboratory through to full production across four mixer sizes (FM-1 through FM-4+), covering vessel volumes from 0.5 liters up to 12,000 liters. The low-positioned mixing plate ensures effective mixing even at reduced fill volumes, which is important during dissolution and formulation steps where heel losses matter.
For process control, the Vibro-Controller VC-201 provides direct adjustment of frequency and amplitude. For facilities requiring full automation, the FUNDAMIX® Vibro-Controller integrates into PLC/HMI and SCADA systems with FDA 21 CFR Part 11 compliance, including audit trail and electronic signature functions.
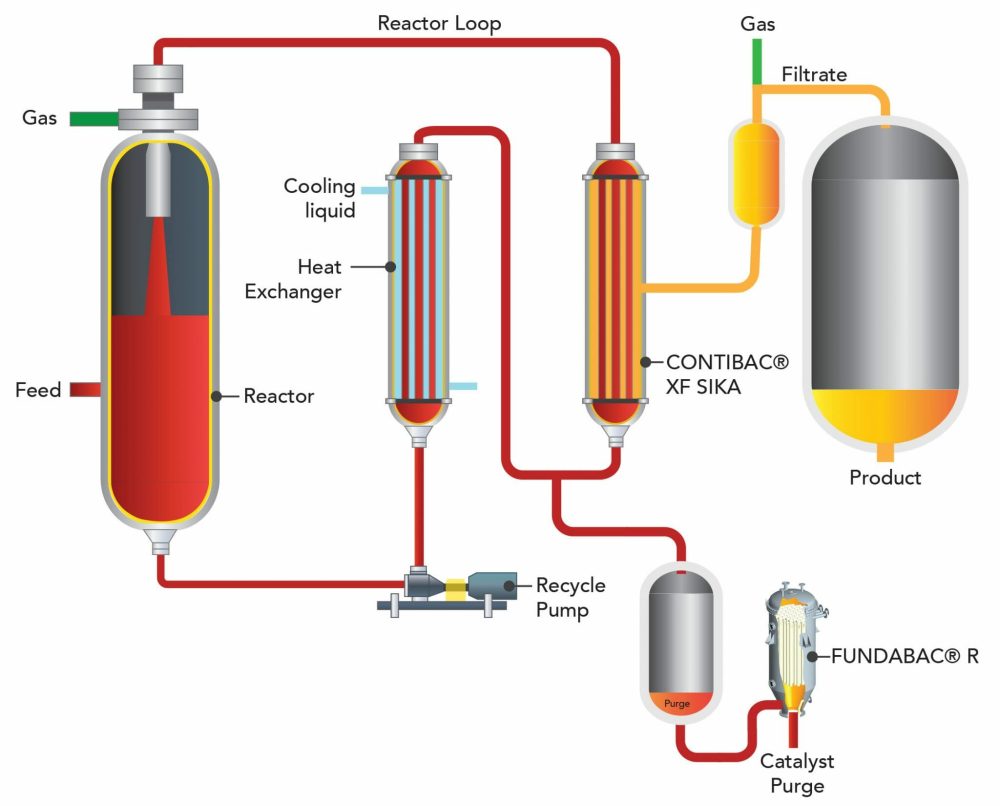
DrM also delivers FUNDAMIX® as complete turnkey plants for insulin manufacturing, combining mixing vessels, CIP/SIP skids, filtration, utilities, and automation into a single factory-tested system. Test and rental units are available for process validation before full-scale investment.
What Makes FUNDAMIX® the Right Choice for Insulin Mixing?
The combination of protein-safe mixing, rapid homogenization, fully sterile operation, and straightforward GMP compliance has made FUNDAMIX® the preferred choice for insulin manufacturers globally. It addresses the core technical challenge of the process: achieving thorough mixing without the mechanical stress events that trigger protein denaturation and batch failure.
The technology is applied across the key mixing steps in insulin manufacturing, including dissolution of the active substance, preparation of formulation buffers, and final dosage solution preparation, each of which places different demands on mixing uniformity and vessel hygiene.
With references spanning insulin, plasma-derived products, vaccines, and other biologics, DrM brings deep application experience to each installation. Lab-scale units allow process teams to validate mixing performance and scale-up behavior before committing to production equipment, reducing risk at every stage of the project lifecycle.
For further information on the FUNDAMIX® Vibro-Mixer and its pharmaceutical applications contact us at [email protected].
References:
Independent academic review: Gimba et al., Vibromixers as an Alternative to Stirred Tanks: Design, Performance Characteristics, and Applications, Chemical Engineering & Technology, 2026.
https://onlinelibrary.wiley.com/doi/10.1002/ceat.70149